 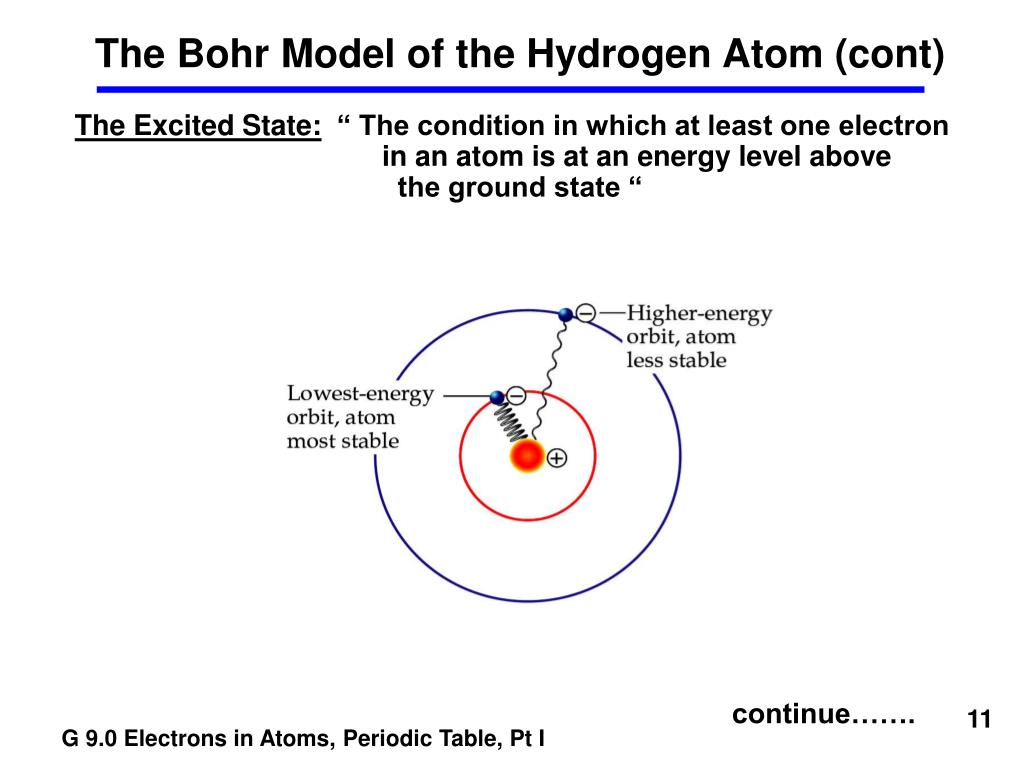
This model works for some aspects of hydrogen, but was found not to fit empirical observations of atoms of other elements. If it spits out a photon, it instantaneously falls down into the lower orbit. If it gains energy by, for example, absorbing a photon (green wiggly arrow), it instantaneously jumps up into the higher orbit. The electron is constrained to specific orbits. In the Bohr visualization of the atom in 1913, the electron circles the nucleus in an orbit like the Earth around the sun. In the simplest form of hydrogen, shown here, it has no neutrons. Hydrogen is the simplest atom and has only one proton (red) and one electron (blue). Physicists call this metaphorical cloud an “orbital.”ĭiagram of the Bohr model of the atom as it applies to hydrogen. A good metaphor for the current understanding of the electron is that it’s like a cloud around the nucleus (see image below). So, almost 100 years ago, physicists threw out the idea of an orbit. However it does not fit the results of experiments with atoms of the other elements nor does it fit later discoveries about the hydrogen atom. 
This model, illustrated by the animation below and to the left, can accurately describe some aspects of a hydrogen atom. This is the solar system model of the atom the electron travels around the nucleus like the Earth around the sun. In the early 1900’s, when physicists were first probing the insides of the atom, they thought that electrons might travel around the nucleus of the atom in an orbit. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
